Introduction
T2DM occurs when insulin secretion is inadequate to meet the increased demand due to insulin resistance then progressively there is apparition of hyperglycemia [1-5]. These children/adolescent are most of the time overweight or obese and commonly are associated either with acanthosis nigricans, PCOS, hyperlipidemia, HTN, and NAFLD. In 75 % of the cases they have strong family history of T2DM [4-5]. The presentation often coincides with the peak of pubertal insulin resistance as explained earlier [8-9].
In contrast to TIDM, children with T2DM may already have microvascular or macrovascular complications by the time they are diagnosed [10]. The differential diagnosis clearly includes T1DM, usually distinguished by the presence of autoantibodies GAD, ICA and IAA; but also diabetes secondary to monogenic causes, transplant and immunosuppression, and other rarer syndromes [9]. The SEARCH for Diabetes in Youth population-based study found that the proportion of T2DM among 10–19 y-olds to vary greatly by ethnicity in the US: 6 % for non-Hispanic whites, 22 % for Hispanics, 33 % for blacks, 40 % for Asians/Pacific Islanders and 76 % for Native Americans [11].
As discussed previously, the average age of T2DM diagnosis in youth is around 14 years, with female predominance. This age of presentation is likely to be related to a time of puberty-mediated insulin resistance in combination with increased body weight. T2DM can be detected while screening asymptomatic children or adolescent because they are belonging to a high-risk population [6, 10].
Some children and adolescents present with diabetes-related symptoms including polyuria, polydipsia, tiredness, blurred vision, vaginal moniliasis, and weight loss [12]. They may also present with acute metabolic decompensation including ketosis, DKA, and HHS but there is no associated islet cell autoimmunity or HLA specificity [12]. Management includes confirming the diagnosis of T2DM; screening for its associated metabolic and vascular complications and initiating lifestyle, dietary and exercise advice to decrease calorie intake and increase energy expenditure combine with metformin or insulin therapy, depending on their glucose control and other risk factors or comorbidities as explained below [10, 12].
Children with T2DM can have three types of clinical presentation: 1). Acute symptomatic: children with T2DM can present acutely with DKA or non-ketotic hyperglycemic coma (NKHC); among children presenting with DKA, 13 % were found to have T2DM; 2). Chronic symptomatic: symptoms are due to hyperglycemia and include polyuria, polydipsia, nocturia and less commonly, weight loss. Adolescent girls may present with vaginal discharge or vulvovaginitis due to monilial infection; 3). Asymptomatic: most commonly, these children are usually identified by routine screening [10, 12]. As mentioned earlier, screening in high-risk groups is recommended to start at the age of 10 years or when puberty starts if it is sooner than that, using FPG every 2 years. The HbA1c target for optimal glycaemic control is less than 7.0%, but the initial target for treatment is negotiated on a mutual agreement depending on their glucose control as discussed below and based on many personal aspects.
Since 95 % of adolescents with T2DM present with obesity, the approach in the counselling of these patients is not much different from the one we use with obese pediatric patients, except for those patients with T2DM that need urgent care. The approach discuss here is the approach mainly recommended in the context of general clinical practice [13-15].
Globally this approach is essentially based on the choice of strategies that will promote the acquisition and maintenance of good lifestyle habits and the motivation to maintain this therapy on the long-term. These strategies will help to change the bad lifestyle habits related to weight gain. Adequate counseling is essential to effectively act on these elements of intervention. To succeed in these tasks, it is highly recommended that family physicians, pediatricians and other HCPs (nurses, dietitians, nutritionists, kinesiologists, psychologists and others) use the “6As” model of counseling that consists of the following mnemonic: “ask, assess, advice, agree, assist, arrange” [13- 15]. This counselling model is use in parallel with the motivational interviewing approach that is very useful to keep the patients in the action and maintenance phases. It is not the purpose of this article to fully detail this approach but mainly to provide the essential information that can be applied in the counseling of T2DM pediatric patients. For more information, please consult the following articles and reviews [13-15].
Ask
In this ‘6 As’ model of counselling, the collaboration of parents is essential and since most children with T2DM first presents with obesity the first think you should do is to ask the permission to talk with the children and their parents about the weight problem of their children [13-15]. Later in the counselling you will have the opportunity to ask questions related to T2DM. Being obese is a delicate issue, because patients and parents are often embarrassed by this condition. It is therefore important not to judge, blame or cause a sense of guilt in patients with weight problems and their parents. You should limit medical jargon and opt for an approach that is sensitive and respectful [13-15].
Asking for permission also allows the HCP identifying barriers and family factors that could have a negative effect on the management of child’s glucose target and child’s body weight. Thus, counseling may be adapted on the basis of these barriers and family factors [13-15]. It is essential that parents are aware of the difficulties, considering that obesity and T2DM are clinical problems which have repercussions on health.
Asking for permission leads to explore attitudes and aptitudes to changes which, among others, are essential to the success of future interventions. Asking permit to determine the perception of children and their parents of their skills to participate in nutritional and physical activity interventions and to make changes in their lifestyle in order to obtain and maintain glucose targets and body weight. It is a way of assessing the availability of the family (children and parents) and social support [13-15].
Ask to assess readiness to change for the patient and his relatives according to the following steps:
- Pre-contemplation: stage where patients and parents have no intention or no interest to change in the near future.
- Contemplation: stage where they start to recognize the reality of the problem and that something needs to be done, but without proposing action;
- Preparation: step where they intend to perform certain actions in the coming months, but nothing concrete is still made.
- Action: step whose time can range from 1 day to 6 months, where they modify their behaviour and their environment to act on the problem;
- Maintenance stage: for a period of more than 6 months, during which they work to reduce relapse and consolidate the gains achieved during the stage of action.
During this stage of the counseling, the HCPs can use motivational interviewing to move patients and parents to the next stages of change; first to the stage of action and possibly in the maintenance stage. The goal of this approach is to bring the child and his parents to make decisions based on their values and their resources, rather than to tell them what to do, as is sometimes the case with the traditional approach [13-15]. The motivational interviewing approach includes the following components:
- Ask the permission to the young and his parents to discuss the issue with the child and their parents. The objective is to provide our support and appropriate information to the patient and his parents.
- Building the report by using active listening, HCPs, seeks to understand the motivations, values and the barriers to change of the patient and his family; the objective is to establish a relationship of trust with them in order to obtain their cooperation.
- Encourage discussion on the changes by guiding the conversation towards the possibility of a change in behaviour; list the positive arguments offered by the patient and his parents and promote other positive arguments. The HCPs must not oppose resistance to the changes proposed by the patient or his parents and should encourage them to propose other solutions, as necessary;
- Guide the conversation towards the realisation of a change and guide the patient and his parents through their change planning in discussing realistic steps and follow-up
Motivational interviewing is a strategy to help patients and their parents to think differently their behaviours and to consider what can be earned by a change. This interview is considered safe and effective for the modification of behaviours associated with obesity [13-15] and is also appropriate for the management of T2DM in pediatric patients. It is based on clarifying expectations, while helping patients and parents to appreciate the values of change and explore the differences between what they think and the reality [13-15]. In addition, motivational interviewing allows building a good patient-doctor relationship, including expressing empathy and congratulating children and parents for good actions towards a positive change in behavior and finding solutions towards failure to achieve appropriate change [13-15].
Assess
Assessment of a child overweight or obese and a child with T2DM should include a complete medical history, a general physical examination, completed by appropriate laboratory tests. Indeed, some children and adolescents present frequently with T2DM-related symptoms including polyuria, polydipsia, tiredness, blurred vision, vaginal moniliasis, and weight loss. The evaluation should include a history of the development of the child, including growth profile, weight gains, compared with the history of growth of parents, a child psychosocial history, including depression, disorders of food, quality of life, self-esteem, as well as a detailed history of risk factor without forgetting the family history of T2DM [10, 12]. The risk factors for the development of T2DM in children discussed earlier should be part of the assessment (see article # 2). Assess if periods are regular, are they painful or heavy, and does the girl suffer from excessive body hair. Consider the risk for obstructive sleep apnoea and assess if there is any nighttime snoring or daytime sleepiness. Other health problems related to obesity include orthopaedic problems such as slipped upper femoral epiphysis; pancreatitis, cholecystitis, and idiopathic intracranial hypertension [13-15].
The medical history should also include the medication list including the, antidepressants and antipsychotic agents and others and those that the mother took during pregnancy, as well as the description of the socio-economic and environmental factors, including home, school and the community environment. The involvement of parents is critical to the assessment of the child and is used to validate the information gathered with the youth [13-15].
Evaluation of nutritional aspects
In this evaluation, it must be relevant to assess for the presence of bad eating habits in children and his family; to assess if the child eats sitting at the family table or in front of the television or in front of other screens; if he eats compulsively, if there is other food problems and if he uses the power of food to manage his emotions [sadness, anxiety, boredom and others] and if the parents use food as a reward [13-15]. Assess also if the child eats food in large quantities, for fun, despite the absence of hunger, and if he feels guilty later. Also assess the consumption of sugary drinks and foods containing added sugars. It is also relevant to assess if the child eats in family or rather in isolation and if he skips meals, especially breakfast [13-15].
Assess with parents for the presence of some habits, such as eating in restaurant in a regular basis and the frequency of meals containing junk food and processed meals [prepared and frozen meals] which often contain a lot of added sugars and salt [13-15]. We must also inquire about the choice of food of the child: is he eating the same thing as the other children in the family? Obviously, in this assessment, it will be recalled that the energy needs of a child may be less or higher than those of the other members of the family. It is essential to assess for the presence of barriers to good nutrition in children and his family, such as the lack of knowledge about the choice, the content, the portions and the cooking, assess with the parents how the child is eating [speed at which meals are eaten, seated at the table, etc.], the ways he resists the temptations and the external pressures promoting excessive intakes, how he deals with the unexpected, the meal planning and, finally, the management of food drives [13-15].
Also assess if the parents know the effects on health of a poor or unhealthy diet, not only in children, but for all members of the family. We must discuss the cost and availability of healthy foods over junk food and understand how personal tastes and culture can affect the choice of food. Finally, we must validate with the child and his parents the information withheld if there were consultations with a nutritionist or a dietitian [13-15].
Evaluation of the level of activity
During this assessment, we must quantify the time allocated to physical activity, i.e. the frequency, duration, intensity, the type of exercise, with which these activities are carried out and finally consider the level of activity in the week compared with the weekend [16]. Also we need to quantify with the young and his parents the time allocated to sedentary behavior, including the time spent in front of the TV or the computer, and the use of video games, phone and other electronic devices. It is important to note the availability for physical activity at home and in school, as well as the obstacles to physical activities. The lack of knowledge, the safety issues and the access to safe equipment, as well as the financial problems in the family that interfere with the regular practice of physical activity should be assessed. Assess if consultations with a specialist in physical activities were performed and analyse the lessons learned from these consultations [13-15]. Finally, it is important to document any conditions that may interfere or be contraindications to regular physical activity in order to adapt the physical activity program to the condition of the patient, as necessary [13-15].
Assessment of psychosocial aspects
Since there is a lot of psychological aspects that we need to take into account in the evaluation of an obese and/or T2DM pediatric patients. It is essential to verify the beliefs of the patient and their parents about the causes and effects of the problem of the child’s, because often these beliefs are based on erroneous perceptions [13- 15]. In addition, do not forget the influences of the cultural and socio-economic problem of the child’s condition. As discussed previously, it is essential to determine the level of preparation for lifestyle changes, the degree of confidence in their ability to make changes as well as their expectations and attitudes towards weight and T2DM management. Also assess the parenting skills; whether parents are authoritarian, or controlling or permissive? It goes the same for family organization: is it a big family, family is disorganized and what is the time available for the child? These elements will help determine the potential for family involvement and the support they can provide to the child [13-15].
Physical examination
It is recommended to include observation of the general appearance, including the severity of obesity and fat distribution in physical examination (peripheral vs. central). Afterwards, you will need to determine the severity of obesity using the BMI. On examination, almost all affected children with T2DM are overweight or obese, with a BMI above the 85th centile for age and sex is defined as overweight while a BMI above the 95th centile for age and sex is defined as obese [13-15]. There is often acanthosis nigricans, a pigmented velvety thickening affecting skin flexures such as the neck, axillae and groins; this is a manifestation of insulin resistance. Blood pressure is often raised (systolic or diastolic blood pressures above 95th centile for age, sex and height. Obviously the physical exam is based on your medical history and should be performed in order to investigate the risk factors and the comorbidities associated with T2DM. Your assessment will be followed by the appropriate laboratory diagnostic tests and those coming from your medical history and physical examination [13-15].
Laboratory testing
A capillary or laboratory plasma or venous glucose is necessary to make the diagnosis of T2DM. It is also helpful to have a baseline HbA1c, to provide an estimation of the duration of hyperglycaemia before diagnosis. In addition to routine measurements should include an assessment for ketone production; urea and electrolytes for assessment of osmolarity and dehydration; assessment for infection [urinary tract, respiratory tract, skin]; autoantibodies to rules out T1DM; and baseline liver function [5].
The C-peptide test is to ensure that the T2DM diagnosis is not being confused with T1DM. C-peptide level is based on blood sugar level and is a sign that the body is producing insulin. A low levels or no insulin C-peptide means that the pancreas is producing little or no insulin. In T1DM, there is a lack of insulin production caused by destruction of β-cells. In T2DM, insulin is produced but the tissues are insulin resistant and in the body therefore there is an increased need for insulin. To combat this, the pancreas produces more insulin but after too long the pancreas loses the ability to produce insulin at all [17]. The other laboratory testing should be performed to assess the complications associated with T2DM.
- Dysplipidemia should be screened at diagnosis of T2DM and every 1-3 years thereafter as clinically indicated. Dyslipedimia should be assessed by measuring the fasting level of total cholecterol, HDL-C, Triglycerides and calculated LDL-C [6].
- Hypertension should be screened at diagnosis of T2DM and at every diabetes-related encounter, thereafter [at least twice annually]. The blood pressure should be assessed by using appropriately sized cuff [6].
- NAFDL should be assessed yearly beginning at diagnosis of T2DM. NAFDL should be assessed by using the ALT and the diagnosis is made when the ALT level is 3 X the normal level [6].
- Nephropathy should be screened yearly commencing at diagnosis of T2DM. Nephropathy should be assessed by measuring the first morning [preferred] or random ACR (albumin to creatinine ratio). An abnormal ACR will require confirmation at least one month later with either a first morning ACR or a timed overnight urine collection for ACR; Repeated sampling should be done every 3 to 4 months over a 6- to 12-month period to demonstrate persistence [6];
- Neuropathy should be screened yearly commencing at diagnosis of T2DM. Questioned and examined for: Symptoms of numbness, pain, cramps and paresthesia: vibration, sense, light touch and ankle reflexes [6];
- PCOS should be screened yearly commencing at diagnosis of T2DM in pubertal females Clinical assessment on history and physical exam should assess for oligo/amenorrhea, acne and/or hirsutism [6];
- Retinopathy should be screened yearly commencing at diagnosis of T2DM. The assessment should include the seven-standard field, the stereoscopic colour fundus photography with interpretation by a trained reader (gold standard); or the direct ophthalmoscopy or the indirect slit-lamp funduscopy through dilated pupil; or the digital fundus photography [6]
Diagnosis of T2DM
According to the American Diabetes Association (ADA) criteria, T2DM is defined as FPG levels of 125 mg/dL (7.0 mmol/L) and above or plasma glucose levels of 200 mg/dL (11.1 mmol/L) and above two hours after an OGTT, while IGT is defined as plasma glucose levels of 140 mg/dL (7.7 mmol/L) and above after an OGTT. In addition to IGT, another prediabetic state has been described: IFG. IFG is defined as serum fasting glucose levels from 100 mg/dL to 125 mg/dL (5.5- 7.0 mmol/L) [5, 17-18].
Epidemiological studies indicate that IFG and IGT are two distinct categories of individuals and only a small number of subjects meet both criteria, showing that these categories overlap only to a very limited extent in children. Recently, the ADA has recommended testing the HbA1c to diagnose T2DM in children. In particular, 6.5% is the lower limit used to diagnose T2DM? This value was chosen on the basis of cross sectional and longitudinal studies conducted in adult subjects showing that a lower limit of 6.5% identifies about one third of cases of undiagnosed T2DM and that subjects have a long term higher prevalence of microvascular complications. Subjects with an HbA1c between 5.7% and 6.4% have been defined as “at increased risk of diabetes” [5, 17-18].
Differential diagnosis of type 2 diabetes in children
- Type 1 diabetes. This is associated with diabetes autoantibodies in about 85% of affected children, and children have an absolute insulin requirement [5, 17-18].
- Apparent type 2 diabetes with coexistent autoimmunity. About 10% of children with an apparent diagnosis of T2DM are found to have antibodies to Glutamate Decarboxylase (GAD), islet cells (ICA), or insulin (IAA). Pancreatic beta cell function is significantly less in antibody positive children and adolescent, and there is more rapid development of insulin dependence. It is likely that these children have T1DM with obesity [5, 17-18].
- Flatbush diabetes. This is seen in some children of African- Caribbean origin, with a strong family history, sometimes autosomal dominant, and with a female preponderance, no HLA association and diabetes autoantibody negative. These children may present with ketoacidosis or ketosis and require insulin initially; but can be weaned off insulin while maintaining relatively good glycaemic control [5, 17-18].
- Monogenic diabetes [formerly Maturity Onset Diabetes of the Young]. This usually presents in families with autosomal dominant history; affects no more than 1% of children with diabetes; is not associated with obesity beyond the prevalence in the background population; and is not associated with insulin resistance [5, 17-18].
Advice
This step leads us to discuss the recommendations of national food guidelines about portions, the variety of food to eat each day, as well as the consumption of foods and beverages low in calories, fat, sugar or sodium. We can emphasize to parents the importance of reading the labels of foods and beverages to make healthier food purchases, to choose products that contain fewer calories, fat, sugar and sodium [13-15]. At this step you can also provide information on the risks to health, which can be reduced by increasing physical activity with or without weight loss and highlight the importance of replacing sedentary activities by physical activity of low to moderate intensity such as using the stairs rather than the elevator or even plan a gradual increase in physical activity for previously sedentary patients, for example starting with short sessions of 5-10 minutes and increasing gradually until the desired physical activity of 60 minutes per day [13-15]. Advice can also note that the management of T2DM is to improve the health and well-being of child and also to reduce and maintain an appropriate management of glucose control as reflected by the number indicated on the glucometer scale [13-15].
Management
The current management plans for T2DM involve lifestyle intervention (LSI) and pharmacotherapy, as necessary [17-22]. The treatment of T2DM requires a family-focused plan delivered by a multidisciplinary team with expertise in dealing with T2DM in children and adolescents. Success in treating T2DM requires addressing the main mechanisms that lead to its development, including insulin resistance and β-cells failure. The multidisciplinary team includes a combination of primary care physicians, pediatricians, endocrinologists, diabetes nurse educators, dietitians, physical activity specialists, social workers, psychologists, and behavioral therapists and may also require the involvement of additional medical subspecialties to address the comorbidities or complications associated with T2DM, as necessary [17-22]. It is important to note that T2DM is associated with other comorbidities that are related to insulin resistance, and some of these comorbidities are present at diagnosis [17-22].
The management plan should be intensive with frequent contacts with family and youth and personalized to the individual patient taking into account the family’s financial resources and being receptive and respectful of ethnic and cultural attributes of the family [13-15]. Engaging the patient and family early and frequently is critical to minimize attrition, which is a common problem in this population. The goals of T2DM management include: 1). achieving and maintaining glycemic control; 2). weight maintenance or weight loss if possible and prevention of weight regain; 3). acquisition of healthy lifestyle habits and skillsets; 4). management of comorbidities, and; 5]. prevention of complications [17-22].
One important study has been published about the role of different treatment modalities in T2DM is the treatment options for T2DM in youth (TODAY) study [23-24]. This was a large, longitudinal, randomized, multicenter study that recruited 699 children and adolescents with an age range of 10–17 years and female to male ratio of 2: 1. These patients were randomized to three treatment groups that included metformin alone or in combination with LSI or rosiglitazone. The mean time since diagnosis of T2DM was 7.8 months and HbA1c less than 8% on enrollment. The primary outcomes, defined as failure to maintain HbA1c less than 8% over 6 months or metabolic decompensation requiring insulin therapy at diagnosis or restarting after stopping insulin within 3 months, occurred in 51.7%, 46.6%, and 38.6% in the above groups, respectively. Metformin alone was no different from metformin plus LSI in improving metabolic outcomes, and higher failure rates in black participants were noted. Combination therapy of metformin plus rosiglitazone offered better success rates especially in girls but was associated with more weight gain [23-24]. This study revealed that, even with intensive LSI and pharmacotherapy, a significant number of T2DM patients fail to achieve adequate glycemic control. In addition, the treatment options available to youth with T2DM are limited when compared to adults, with insulin and metformin being the main agents used. This emphasis the need to find solutions for the development of clinical trials testing new molecules adapted to pediatric patients presenting with T2DM. This issue will be discussed in depth in the articles number 5. Similarly with the development in the use of pharmacogenomics and the pharmacokinetic, it will become possible to individualize therapy for the treatment of T2DM; to offer treatment that will be more efficient to a specific individual with less adverse drug reactions. This issue will be further discussed in the article number 6 of the current issue.
The risk of microvascular and macrovascular complications in adults increases with both the duration of T2DM and lack of glycaemic control, so it is vital to achieve and sustain metabolic control through normalization of glycaemia; and control of co-morbidities. Therefore, reducing the risk of microvascular and macrovascular complications may require even tighter and longer glucose control in childhood with T2DM than in adults [23-24].
Life style intervention Program [LSI]
The main emphasis in the management of T2DM is on lifestyle modification [20-22]. LSI defined as an increase in physical activity, decrease in sedentary activities and a change in dietary patterns that result in a daily caloric deficit, is considered the main component in the management of T2DM and its-associated complications in pediatric patients [20-22]
LSI works best when the whole family is engaged in learning process about supporting the child, and these recommendations may be helpful for other family members who may suffer from obesity and but not still diagnosed T2DM [13-15]. Parents and caregivers need to be informed of the importance of modeling healthy behaviors in a way to encourage children to acquire appropriate lifestyle habits [13- 15]. They also need to maintain positive reinforcement of success and avoid penalizing failures. The key is to build collaborative relationships and avoid combative interactions when it comes to managing T2DM with their child [13-15]. The present section examines the lifestyle management efforts to prevent and treat T2DM and its associated co-morbidities in children.
Nutrition
Regarding nutrition, the focus should be on formulating a nutrition plan that involves food composition and eating behaviors that reduce excess caloric intake [13-15]. Recommendations regarding controlling portion size and setting up regular meal and snack times are important. Equally important is eating meals as a family and elimination of distractions during meal times including TV, computers, or other disturbances to slow down eating and improve social interactions [13-15].
Other recommendations include avoiding snacking especially while watching TV, using the computer, and late at night. In addition, the elimination of sugary drinks and foods with high fat or caloric content is also critical in reducing caloric intake in order to promote weight maintenance or weight loss and better promote glucose control.
Parents, caregivers, and youth need to be taught how to read food labels to understand the nutritional value of consumed foods and to emphasize the importance of consuming less fat including saturated fatty acids, increasing fiber intake, and reducing sugar intake and eating out in restaurant or eating prepared food that contains a lot of sugar and salt [13-15].
The most prevalent goal is to stop weight gain in order to obtain better glucose control. The focus may not always be on caloric restriction as that may interfere with growth but the focus should be on keeping the blood glucose and HbA1C within the normal range. This can be done through lifestyle changes such as decrease of high calorie and high fat foods, as well as foods high in simple carbohydrates, especially sugar [13-15].
Nutrition education should be provided in a way that encourages regular meals and regular healthy snack each day, as well as regular physical activity. Total carbohydrate consumption will need to be monitored as recommended by the diabetes nurse. These carbohydrates should be complex carbohydrates [esp. legumes, fruits, vegetables, and oats] that help the patient increase his fiber intake as it takes the small intestine longer time to absorb high fiber foods and therefore affects less glucose levels.
Because many patients with T2DM their kidney is not functioning properly; as demonstrated by the presence of protein in their urine, they will have to be careful not to consume more than 0.8 g/kg or ~10% of total kcal at least until there is no longer protein present in their urine and then the patients should consume no more than 20% of their total kcals from protein. Their total fat consumption should not exceed 25-35% of total kcal and saturated fat should not be higher than 7% of total kcals [13-15].
The Committee [Consensus statements of Focus on a Fitter Future on T2DM prevention using diet and physical activity] supports dietary intervention to manage weight, specifically a low glycemic index [GI] diet implemented by registered dietitians, to influence metabolic risk factors for T2DM in pre-diabetic children. More-severe carbohydrate restriction may be considered for severely obese children with pre-diabetes under medical supervision [25]. Reducing sugar-sweetened beverages (SSBs) intake in children and adolescent has been shown to have a positive impact on weight. Plourde [13-15] and others go even further on this issue by recommending “0” SSB or added sugar to overweight and obese children. Please also note that the simple slogan “5, 3, 2, 1, 0” discussed in the prevention section is also applicable in the treatment approach of pediatric T2DM.
Intensive dietary interventions without adjunct exercise therapy elicit a ~2 % (95%CI −2.40 to −1.23 %) weight loss defined as weight, BMI, % body fat, waist circumference or skinfold thickness, relative to controls receiving standard dietary recommendations. In a recent position statement from the Academy of Nutrition and Dietetics, four dietary strategies for eliciting weight loss in overweight and obese children and adolescents were recommended: (1) modified traffic light diet, (2) low carbohydrate diet, (3) reduced glycaemic load (GI) diet, and (4) non-diet approach. Systematic reviews have determined that these strategies are effective in improving body composition in the short term among overweight and obese children and adolescents [25].
Physical Activity
According to the 2008 Physical Activity Guidelines for Americans, “being physically active is one of the most important steps that Americans of all ages can take to improve their health”. Children and adolescents should engage in 60 min daily of moderate-to-vigorous PA (MVPA). The recommendations by the American Academy for Pediatric and the World Health Organization are essentially identical. Current data suggest that only 8% of U.S. youth meet the 60-min/day MVPA recommendation, based on accelerometer data, and that many children and adolescents exhibit sedentary behavior and are subsequently largely physically inactive. A similar picture is also observed in our youth in Canada [26].
We should focus on the health risks of sedentary behavior because physical inactivity has been identified as the fourth leading risk factor for global mortality. Therefore, it is essential that more information be obtained regarding physical activity and sedentary behaviors in T2DM youth in order to be able to intervene on the main sedentary behaviours as demonstrated in a recent article by Plourde [16]. The beneficial effects of increased physical activity and decreased sedentary behavior are extremely important in youth with T2DM because of the markedly increased long-term risk of cardiovascular disease in this population compared to persons without T2DM [13-15, 27].
The independent role of exercise training without caloric restriction on reducing insulin resistance is recently recognised as an adjunct in the management of T2DM in children. In fact, some recent studies observed that both the aerobic and resistance type of exercise training without calorie restriction resulted in meaningful changes in insulin sensitivity, suggesting that exercise alone is an effective therapeutic strategy in overweight and obese youth. As observed in adults, this beneficial effect occurs through multiple adaptations such as improved glucose uptake of skeletal muscles and body composition changes in overweight children and adolescents [13-15, 27-28].
Physical activity and cardiorespiratory fitness in children and adolescents are both correlated with insulin sensitivity independent of adiposity, especially when physical activity is at higher intensities. Fedewa and colleagues conducted a meta-analytic review to determine the effect of exercise training on predictors of T2DM in children and adolescents. They found small to moderate effect sizes for exercise training on fasting insulin providing support for the inclusion of physical activity in lifestyle management programs to prevent and treat T2DM in youth [29]. Taken together, results from these systematic reviews reveal that intensive, structured lifestyle interventions, particularly those that include both exercise and dietary modifications, yield modest but meaningful improvements in adiposity in obese children and adolescents [13-15]. These data suggest that similar strategies may also be beneficial for obese youth living with T2DM. Among overweight and obese youth, lifestyle interventions confer favourable effects on serum lipoprotein profiles, fitness, insulin sensitivity and systolic blood pressure. These results explain why often it is not necessary to introduce medication to treat dyslipidemia and/or HTN in pediatric or adult patients presenting T2DM as physical activity and modest weight loss are often sufficient to correct these T2DM-associated disorders [13-15, 28].
A) Intervention Techniques:
Techniques taught to children and adolescents as well as family members include self-awareness, goal setting, stimulus control, coping skills training (CST), cognitive behaviour strategies and contingency management. Importantly, parents played a key role in the intervention and were taught to play role modelling of healthy behaviours and coping strategies. In the recent book and review articles by Plourde [13-15] there are good examples on how to use these techniques with youth presenting weight problems and their family members? Studies demonstrate that these techniques are associated with significant reductions in BMI, body weight, body fat and percent body weight and fat. The improvement in body composition was associated with significant improvements in total cholesterol and fasting insulin. These effects also translated into an increased rate of remission from IGT in obese children with abnormal glucose levels. The effects of these interventions techniques, suggest that under ideal conditions, clinically relevant weight loss and positive metabolic health outcomes are achievable and sustainable in obese children and adolescents by using these techniques [13-15].
B) What are the established barriers to exercise participation?
Children and adolescents with T2DM experience similar exercise barriers than those who are overweight or obese, such as lack of time and motivation, inability to access facilities and others [13- 15]. These individuals are most of the time are overweight or obese and may feel that the benefits of exercise do not provide a sufficient gain and commonly report physical discomfort, boredom, and lack of time as the major barriers to exercise [13-15]. Similarly, children and adolescents with T2DM are more likely to be discouraged from exercising because of physical discomfort. Body-related concerns, such as being seen by others while exercising, are also frequent barriers to exercise, particularly in overweight girls [13-15].
Another factor to consider with regard to exercise participation in both children and adolescents is peer influence. With peer presence, some studies suggest children are able to increase participation by 54%, however negative peer perceptions may also have a negative impact on exercise in those children and adolescent. Given the social stigma associated with being overweight or obese, and the perceived negative body image barrier of adolescents, peer influence may impede obese individuals from exercising [13-15]. Importantly, recent data also suggests adverse exercise kinetics may be a significant barrier to sustaining an exercise program in children and adolescents with T2DM. Nadeau et al showed that children and adolescents with T2DM have decreased maximal oxygen uptake (VO2 peak), lower maximal work rates, and significantly prolonged VO2 kinetics compared with obese non-diabetic controls. Thus, it appears that exercise at the required intensity for health benefits may be inherently more difficult, uncomfortable, and the metabolic adaptations to intense effort slower, resulting in greater levels of overall discomfort for those with T2DM [30].
C) Strategies to overcome barriers to participation.
Adolescents with T2DM may be relatively unconcerned about long-term consequences of poor metabolic control. Also, these youth often have other family members with T2DM, and modeling of good exercise habits may thus be problematic within the household family unit. Therefore, it is important for both youth and parents to be aware of and understand the long-term consequences of poorly managed T2DM.
Support from parents and family members are also critical for encouraging exercise in children and adolescents with T2DM. When children are given action-oriented support, rather than verbal prompts, they are more likely to be active [13-15]. Not only should families engage in activities with children and adolescents, but they should also help the child overcome body-related barriers [13- 15]. It is important to help youth understand the benefits of exercise and establish concrete realistic goals. Additionally, teaching those that are overweight or obese to reduce the value of esthetics can help reduce social pressures and improve body-esteem, could help them to better adhere to an exercise prescription.
For adolescents specifically, support from peers with T2DM could be beneficial. Adolescents may find it very helpful to share and learn from experiences of other adolescents with T2DM, and the absence of peers may negatively influence management. Along with support and education, it is important to consider the child’s interests and capabilities. It is important to be aware that enjoyment is a key and developmentally important factor in exercise for children and adolescents, and may improve adherence to exercise. Self-efficacy should also be considered to avoid setting unattainable goals that can diminish adherence to physical activity programs [13- 15]. Therefore, as in overweight or obese children and adolescents, individual behavioral approaches with family support should be emphasized to facilitate exercise in youth with T2DM.
Sedentary behaviors
Increased screen time is associated with increased sedentary time and obesity. While there are no specific guidelines to address screen time use in T2DM, the recommendations for prevention of childhood obesity by the American Academy of Pediatrics is limiting screen time to two hours per day excluding use for academic purposes or for work and seem reasonable to follow in T2DM [13-15]. Sustaining LSI is a challenge in the T2DM population, and in one study only 17% of patients have lowered their BMI over 1 year of LSI implementation, and 23% were off medications over 2 years. LSI is essential not only to manage T2DM per se but more so to deal with its associated complications including fatty liver disease and to modulate future CVD risk [13-15].
Electronic media use in T2DM youth seems to be very high with on average 3.6 h for boys and 2.9 h/day for girls, of which the majority is spent watching TV. Rothman et al. reported that only 32% of their population watched one hour of TV or less per day, while the remained watched 2 or more hours [31]. Compared to youth without diabetes, those with T2DM seem to engage in markedly more sedentary behaviors. Minimizing sedentary behavior during waking hours is likely beneficial for the prevention and management of T2DM in the pediatric population. Evidence suggests that ~80% of waking hours during the preschool years are spent sedentary [31]. Therefore, there is a lot of room for reducing this component of LSI and increasing physical activity to improve the management of pediatric patients with T2DM.
Pharmacotherapy
LSI is important to provide the basis for acquiring healthy lifestyle habits in T2DM. Even though, the success rates of maintaining glycemic targets based on LSI alone is important, often starting pharmacotherapy at diagnosis is appropriate [19, 32-36]. The aims of pharmacological therapy are to decrease insulin resistance (e.g. metformin), increase insulin secretion [e.g. sulphonylureas, not recommended in children], slow postprandial glucose absorption [acarbose, not recommended in children], or finally to increase glucose entry into cells (insulin). For children, the choice is limited to insulin and metformin as these are the only two molecules authorised for the treatment of T2DM in pediatric patients [6].
Metformin
Metformin is now considered as first line oral antidiabetic drug (OAD) in pediatric population with T2DM by ADA (American Diabetes Association) and ISPAD (International Society of Pediatric and Adolescent Diabetes) [17-18]. US-FDA has approved metformin in children above 10 y of age. It is a biguanide that lowers blood glucose levels via several mechanisms including: i). reducing hepatic glucose output by inhibiting gluconeogenesis; ii). increasing insulin-stimulated glucose uptake in muscle and adipose tissue; iii). inhibiting inflammation in cells by inhibiting the NFκB pathway which, when active, interferes with insulin signaling; iv), increasing fatty acid oxidation in muscle and inhibiting fatty acid synthesis in fat and liver by upregulating AMPK activity; and v) enhancing the secretion of GLP-1 from the gut [19, 32-36]. Metformin has an initial anorexic effect and may result in modest weight loss. It lowers HbA1c by 1-2% and is to be taken with food to minimize its gastrointestinal (GI) adverse drug reactions including nausea, vomiting, diarrhea, and abdominal pain [19]. There are slow release preparations such as Glucophage XR and others that have less GI side effects and are taken once daily, which may improve compliance, and pediatric trials are ongoing to evaluate their efficacy.
Importantly, metformin use is rarely associated with hypoglycemia. GI adverse events are common with metformin but they can be minimized by taking the OAD after meals, slow titration of doses or by the use of extended release preparations. Titration of metformin is done as follows: start with pills of metformin 250 mg once a day for 3 to 4 days and if tolerated, increase to 250 mg twice daily. The dose is slowly titrated upwards by 500 mg per week over a period of 3 to 4 week to a maximum of 2000 mg/day, given as two divided doses or as single dose of sustained release preparation. Metformin reduces HbA1C by 1–2 % [19]. Metformin should be avoided in children with severe renal impairment, hepatic dysfunction, or cardio respiratory dysfunction due to the risk for lactic acidosis [19]. However, the risk of lactic acidosis is relatively rare. Most HCPs treat patients with new-onset T2DM who are asymptomatic, have HbA1c less than 9%, or have blood glucose concentrations in the low to mid-200s with metformin before initiating insulin treatment [17-18].
The results of the TODAY study demonstrated that the rates of treatment failure include HbA1C ≥ 8.0 % or metabolic decompensation were 51.7 % with metformin alone, 38.6 % with metformin plus rosiglitazone, and 46.6 % with metformin plus LSI. Metformin plus rosiglitazone was associated with a 25.3% decrease in treatment failure as compared with metformin alone (P=0.006); the outcome with metformin plus LSI was intermediate but did not differ significantly from the outcome with metformin alone or with metformin plus rosiglitazone [23]. But it is essential noting that the rates of failure observed with metformin alone or with metformin plus LSI are rather high.
In the review made by Scheen AJ [37], it has been shown that metformin is carried into the hepatocytes by an organic cation transporter 1 (OCT1), which is encoded by the gene SLC22A1. The data from animal and human have demonstrated that OCT1 is important for the therapeutic action of metformin. These data also indicated that the genetic variation in OCT1 may contribute to variation in response to the OAD [38]. The effects of metformin in OGTT were significantly lower in individuals carrying the reduced function polymorphisms of OCT1. Therefore, it is possible to explain that the rate of failure observed in the TODAY study might be explained in part by this genetic variation in OCTI [37].
Severe metformin intolerance has also been associated with this reduced function of the OCT1 variants. In the GoDARTS study, GI intolerance to metformin has been observed four times more frequently in individuals with the two reduced-function of the OCT1 alleles who were treated with the OCT1 inhibitors [39]. These results, which were confirmed in another study, suggest that high inter-individual variability and the severe GI intolerance to metformin shares a common underlying mechanism [40]. These data seems to suggest that patients carrying these genetic variations in the OCT1 respond less to metformin and are more associated with GI adverse drug reactions. Which means that the identification of this genetic variation before initiating the treatment with metformin could contribute to a more personalized and safer metformin treatment [40]? Finally, genetic variants associated with metformin response could be used to predict both the glucose-lowering efficacy and tolerance profile of metformin treatment in patients before they take the drug, a step forward in the path towards personalized medicine [41-42]. The latter concept will be further discussed in the article number 7 of the current issue.
Failure of metformin as monotherapy indicates the need for addition of insulin. The goals of therapy should be to achieve HbA1C
Insulin
It cannot be stressed enough that if there is any doubt about the diagnosis of T2DM, then it is much safer to commence insulin treatment and revise the diagnosis later [35-36]. As you will see in the article number 5 this approach complicates the eligibility of pediatric T2DM patients in participating in clinical trials having the goals of finding more efficient new OAD to treat this complicated metabolic disorders.
Insulin reduces islet glucotoxicity and has a paradoxical effect on improving insulin sensitivity in the context of insulin resistance. Insulin is used at presentation if the patient is hyperglycemic including blood glucose > 11.1 mmol/L, ketotic, or ketoacidotic or if the HbA1c is ≥ 9%. The goal of insulin therapy is to reverse the acute metabolic decompensation noted in some patients at presentation and may be used for few weeks at diagnosis and then is withdrawn gradually thereafter [35-36].
In some cases, it is not possible to de discharged from insulin. In addition, insulin is usually needed for few years after the initial diagnosis on a maintenance basis due to β-cell failure. There are few pediatric data on the insulin regimens used, but they all seem to have equal efficacy [35-36]. Insulin may be the only prescribed agent in pediatric patients with T2DM. The main side effects of insulin include weight gain and hypoglycemia; two major adverse effects that need close monitoring.
Other Medications
There are limited data on the use of additional OAD to treat T2DM in pediatric patients compared to adults. Amylin (pramlintide), incretinmimetics/ glucagon like peptide-1 receptor agonists (exetanide), DPP-1 V inhibitors (vildagliptin, sitagliptin, saxagliptin, linagliptin) and á-glucosidase inhibitors are not approved in patients
Sulfonylureas [SU]
Sulfonylureas are insulin secretagogues and therefore are only effective if residual pancreatic insulin secretion is present. They bind to the sulfonylurea receptor on the β-cells; this results in closure of the KATP channel and depolarization of the cell membrane and then calcium influx through the calcium channels, which results in insulin release [19, 32-36].
From a physiologic point of view, the clinical response to sulphonylurea has been widely associated with a number of gene polymorphisms, particularly those involved in insulin release [37]. The hepatocyte nuclear factor- 1 alpha (HNF1-α) gene mutations are the commonest cause of monogenic diabetes. Diabetic patients with HNF1-α gene mutations are particularly sensitive to the glucose-lowering effect of sulphonylureas and therefore patients presenting these mutations can respond more efficiently to this OAD [43].
As far as T2DM is concerned, genetic markers of genes that predict treatment outcomes of sulphonylurea therapy have been recently reviewed: especially, the ABCC8 (SUR1), KCNJ11 (Kir6.2), TCF7L2 (transcription factor 7-like 2), and IRS-1 (insulin receptor substrate-1). A convincing pattern for poor sulphonylurea response was observed in Caucasian T2DM patients with the rs7903146 polymorphisms of the TCF7L2 gene [44-45]. Another example is the Arg (972) insulin receptor substrate-1 (IRS-1) variant which is associated with an increased risk for secondary failure to sulphonylurea [46].
In addition to the effects of genetic variants on target genes, variation in the enzymes responsible for sulphonylurea metabolism also can affect the OAD efficacy [37]. An increased sensitivity to sulphonylurea, with a potential higher risk of hypoglycaemia, has been reported in T2DM patients with reduced function alleles at CYP2C9, resulting in a reduced metabolism of the OAD [47]. As a consequence, the total oral clearance of all studied sulphonylureas [tolbutamide, glibenclamide (glyburide), glimepiride, glipizide] was only about 20% in persons with the CYP2C9*3/*3 genotype compared with carriers of the wild type genotype CYP2C9*1/*1, and the clearance in the heterozygous carriers was between 50% and 80% of that of the wild type genotypes [48]. However, the resulting differences in sulphonylurea-associated glucose-lowering effects were much less pronounced. Nevertheless, CYP2C9 genotype-based dose adjustments derived from PK studies may reduce the incidence of adverse reactions, especially hypoglycaemia [48]. However, because of important limitations in available studies, further studies are necessary before developing personalized medicine for T2DM management with sulphonylurea [49].
Thiazolidinediones [TZD]
Rosiglitazone and pioglitazone are the only remaining OADs from TZD family in clinical use, and rosiglitazone was used in the TODAY study [23]. Rosiglitazone binds to peroxisome proliferator-activated receptor gamma [PPAR-γ] in metabolic cells. This is a transcription factor and master regulator of fat and carbohydrate metabolism and is an insulin sensitizer. In adults, rosiglitazone reduces HbA1c by 0.5–1.3% [19].
From a biologic point of view, CYP2C8 and CYP3A4 are the main enzymes catalyzing the biotransformation of pioglitazone, whereas rosiglitazone is metabolized by CYP2C9 and CYP2C8 [48, 50]. Please note that troglitazone, a TZD has been withdrawn from the market because of hepatotoxicity. The genes coding for the CYP2C8 and the PPAR-γ are the most extensively studied to date and the selected polymorphisms may contribute to the respective variability in the pioglitazone pharmacokinetics and pharmacodynamics, which may impact both the efficacy and toxicity of the OAD [51]. The CYP2C8*3 polymorphism was found to be associated with lower plasma levels of rosiglitazone and thus with a reduced therapeutic response but also to a lower risk of developing oedema. These observations suggest that the individualized treatment with rosiglitazone on the basis of the CYP2C8 genotype may therefore be possible [52]. However, the studies that looked at the association between CYP polymorphisms and TZD toxicity were inconsistent and generally did not produce statistically significant results [53].
Specific genetic variations in the genes involved in the pathways regulated by TZDs could also influence the variability in the treatment with these OADs [54]. A first study showed that the Pro12Ala variant in the PPAR-γ gene does not affect the efficacy of pioglitazone in patients with T2DM, suggesting that the glucose-lowering response is independent from the pharmacogenetic interactions between PPAR-γ and its ligand pioglitazone [55]. However, in a more recent meta-analysis, which synthesized the currently available data on the PPAR-γ Pro12Ala polymorphism, the carriers had a more favourable change in fasting blood glucose from baseline as compared to the patients with the wild-type Pro12Pro genotype [51].
In a study investigating the influence of the S447X variant in the lipoprotein lipase [LPL] gene on the response to therapy with the TZD pioglitazone, the S447X genotype conferred a statistically significant reduction in the glucose-lowering response rate to pioglitazone as well as a less favourable lipid lowering response relative to the S447S genotype [56]. In a study in Chinese patients with T2DM, the adiponectin gene polymorphism rs2241766 T/G was associated with a higher pioglitazone efficacy [57]. Therefore, pharmacogenomics and pharmacogenetics studies may be important tools in drug individualization and therapeutic optimization when prescribing TZDs in patients with T2DM [54]. However, progress still remains to be made before more evidence becomes available in children. This important issue will be further discussed in a following article.
Incretin Mimetics
This class of OADs includes glucagon-like peptide-1 (GLP-1) receptor agonist, which is a peptide secreted by the L cells of the small intestine in response to food, and has a half-life of 2 minutes [32], making it impractical for clinical use. It enhances insulin secretion in response to glucose, suppresses glucagon production, delays gastric emptying, prolongs satiety, and reduces HbA1c and weight. It is given subcutaneously twice daily, which may limit compliance in teens. Some of its side effects include nausea, vomiting, diarrhea, dyspepsia, and headache [19].
Studies are ongoing to validate its use in T2DM pediatric patients. Incretins are gut hormones that were originally discovered because studies of insulin release demonstrated greater insulin release to oral glucose than to glucose given intravenously. It was then postulated that this difference in insulin secretion was secondary to the effect of hormones produced by the gut and to the effects of incretins. Although there are several incretins, only three of them, the gastric inhibitory peptide (GIP) and the glucagon-like peptides 1 and 2 [GLP- 1 and GLP-2] are strong insulin secretagogues [32].
GIP also stimulates glucose uptake by adipocytes, thus enhancing lipogenesis. Although all three of these incretins can improve glucose homeostasis, the late phase insulin response to GIP was found to be absent in individuals with T2DM. GLP-2 is an epithelial growth factor, so studies have focused on GLP-1 as a potential therapeutic OAD. GLP-1 is secreted by the L cells of the distal ileum largely in response to carbohydrate and fat, which act directly on the L cells to stimulate GLP-1 secretion. This in turn results in insulin secretion and in a concurrent decrease in plasma glucose concentrations. Studies in adolescents have shown that these drugs have helped them to lose weight [32] which is essential for reducing insulin resistance in patients with T2DM. The most common adverse drug reaction associated with this drug is pancreatitits [19]. This underscores the need for continuous monitoring for serious adverse events with the use of this OAD as it has only been in use for a relatively short time. This serious adverse drug reaction would certainly limit its development for the treatment of T2DM in pediatric patients.
The most commonly used GLP-1 agonists are exenatide (Byetta), given twice daily with meals, and liraglutide (Victoza), a once daily analogue [19]. In order to attempt to improve adherence with an injectable medication in patients with T2DM, a long-acting analogue (LY2189265) that can be given once weekly has been developed. Studies of pharmacodynamics of escalating doses of LY2189265 in six patients indicated increase in glucose dependent insulin secretion and decreased glucose excursions during an OGTT at all doses studied (0.1– 12 mg), but there was an increased pulse rate with doses of at least 1.0mg and an increased diastolic blood pressure with doses of at least 3.0 mg [58]. GI adverse drug reactions also increased with increasing doses, indicating that the low dose, but not the high dose, appears to be well tolerated and effective. Further research are needed before these OADs be authorised in pediatric patients.
DPP-IV Inhibitors
These OADs inhibit dipeptidyl peptidase- (DPP-) IV, the enzyme that degrades incretin hormones. DPP-IV inhibitors thus increase the activity of endogenous GLP-1 [32]. They do not affect gastric emptying, satiety, or weight; issues that are important for youth with T2DM, since most of whom are overweight or obese. They are given once daily orally with metformin, and there are no pediatric data to evaluate their role in T2DM.
Amylin
This is a peptide released with insulin by the β-cells at a ratio of 1: 100. It reduces glucagon production, slows gastric emptying, and reduces food intake. It is given to patients who are on insulin and causes a mild reduction in the HbA1c and mild weight loss but is associated with nausea and hypoglycemia that requires the reduction of insulin dosing by as much as 50% [32]. There are no data on its use in T2DM children and adolescents. Amylin, is an islet amyloid polypeptide co-secreted with insulin by the β-cells of the pancreas [19].
Pramlintide acetate (Symlin) was approved by the FDA in 2005 for adults with T1DM or insulin-dependent T2DM. It is given subcutaneously prior to meals with insulin. The most clinically significant risk is hypoglycemia, especially in individuals with T1DM. Side effects are mainly GI, including nausea, anorexia, and vomiting. In the late 1990s and early 2000s pramlintide acetate was demonstrated to improve glycemic and weight control through the control of postprandial glucose excursions in adults with T1DM and insulin- dependent T2DM [32]. But this molecule has not been approved yet in children and adolescents for the treatment of T2DM.
Alpha Glucosidase Inhibitors
This class of OADs delays the absorption of carbohydrates by inhibiting the breakdown of oligosaccharides in the small intestine. It can reduce HbA1c by 0.5–1% [19]. It needs to be taken before every meal, and flatulence is a side effect [19]. Both of these reasons make them less desirable for the T2DM teen.
Bariatric Surgery
In adults, bariatric surgery results in the remission of T2DM and discontinuation of medications in many situations [6]. In the only pediatric study published so far, 11 adolescents with T2DM who underwent Roux-en-Y gastric bypass were compared to those who were medically managed; the surgical group had around 34% reduction in BMI and improved their control of HTN and dyslipidemia. In 10 patients from the surgical group, T2DM disappeared and they did not require pharmacotherapy [59]. The experience with bariatric surgery in adolescents with T2DM is very limited with specific eligibility criteria including BMI >35 kg/m2; Tanner stage IV or V, and skeletal maturity. Furthermore, all other approaches must be taken first, primarily the initiation of a healthy lifestyle that includes a healthy diet as well as exercise for a period of 6-12 months [13-15]. As this is a new procedure in T2DM teens, longitudinal studies are needed to validate its feasibility.
Extremely obese diabetic adolescents experience significant weight loss, remission of T2DM, improvements in insulin resistance, improvements in beta-cell function, and CVD risk factors after Roux-en-Y gastric bypass surgery [60]. Although the long-term efficacy of Roux-en-Y gastric bypass is not known, these findings suggest that it is an effective option for the treatment of extremely obese adolescents with T2DM. In RCTs of treatment with the gastric banding procedure vs. a lifestyle weight loss program for adolescents with severe obesity, more than 50 % weight loss was achieved with complete resolution of the metabolic syndrome and insulin resistance with the surgical approach and this effect was sustained over 2 y of follow-up after the surgical procedure [60-61].
Management of Comorbidities/Complications
It is not the purpose of this section to discuss the management of all the complications associated with T2DM and the discussion will be limited to the most common ones that include hypertension, renal impairments and dyslipidemia. The readers can have access to further information concerning the management of other comorbidities by consulting previous documents by Plourde [13-15].
Hypertension
Screening for high blood pressure should begin at the time of diagnosis of T2DM and continue to be taken at every diabetes-related clinical encounter thereafter, since up to 36% of adolescents with T2DM have HTN. The Treatment of high normal blood pressure i.e., systolic or diastolic blood pressure consistently above the 95th percentile for age, sex and height should include LSI to reduce weight. If target blood pressure is not reached after 3 to 6 months of LSI, pharmacologic treatment should be considered. ACE inhibitors are teratogenic and should be cautiously used in sexually active adolescent girls. The goal of treatment is a blood pressure <130/80 or below the 90th percentile for age, sex and height whichever is lower. Combination therapy may be required if HTN does not normalize on single agent. If ACE inhibitors are not tolerated angiotensin receptor blocker (ARB) can be considered. [6, 17]. Most of the time minimal weight loss and adherence to LSI is sufficient to correct HTN in overweight or obese T2DM patients.
Renal Impairments
Urine albumin at concentrations >30 mg/g creatinine should be considered as a continuous risk marker for CVD events. Microalbuminuria should be treated with an ACE inhibitor or if not tolerated, ARB similar to the management of HTN. Combination therapy may be required if albuminuria does not normalize on single agent. In patients with T2DM, HTN and microalbuminuria, both ACE inhibitors and ARBs have been shown to delay the progression of macroalbuminuria In patients with T2DM, HTN, macroalbuminuria, and renal insufficiency (serum creatinine >1.5 mg/dL), ARBs have been shown to delay the progression of nephropathy [6, 17]. The presence of renal impairment should be considered when selecting both the type and the dose of oral glucose-lowering agents in patients with T2DM [19]. More particularly, this is the case for metforminm, incretin-based therapies (DPP-4 inhibitors and GLP-1 receptor agonists) and SGLT2 inhibitors. The risk of hypoglycaemia is also increased in T2DM patients receiving sulphonylureas in the presence of renal insufficiency [19].
Dyslipidemia
Children with T2DM have an increased prevalence of dyslipidemia with 44.8% of Canadian children reported to have dyslipidemia at the time of diagnosis [6]. Testing for dyslipidemia should be performed soon after the diagnosis when blood glucose control has been achieved and annually thereafter. The goal is LDL-cholesterol 1.9 mmol/l. In children with familial dyslipidemia and a positive family history of early CVD events, a statin should be started if the LDL-C level remains >4.1 mmol/L after a 3- to 6-month trial of LSI. As for HTN, most of the time minimal weight loss and adherence to LSI is sufficient to correct dyslipidemia in overweight or obese T2DM patients.
Mutual agreement
We are making progress in terms of how we are treating T2DM. We are now going for a more individualized approach that is why this component of the “6As” model of counselling is so important. Through our understanding of some of the pathophysiology of T2DM, disease and patient’s characteristics we are now able to be more patient-specific on treatment targets. Specifically, the ADA has put forward a very nice graphical representation of individual physiologic and patient-centered aspects [https://durobojh7gocg.cloudfront.net/ content/diacare/38/1/140/F1.large.jpg] that one should incorporate in the selection of the treatment target that we can then negotiate with the patient in the ‘agree’ step of this “6As” model of counselling. This is not something that takes a long time. When we sit with a patient, it takes only few minutes to discuss some of their disease characteristics such as their risk for hypoglycemia, how long they have had the disease, whether they have other important comorbidities, their risk of weight gain, their motivation status. Based on this information, you can easily achieve a mutual agreement on an initial treatment target HbA1c goal. I recommend that you put it in the patient’s chart so that other HCP who see the patient will have a perspective and a rationale for why you are approaching treatment the way you do [13-15].
Again, since for most of the patients with T2DM are overweight or obese, it is agreed that obtaining an ideal BMI is not a realistic goal for the majority of children and adolescents with T2DM. Considering that non-realistic goals may lead to failure [13-15], one of the first objectives should be to stabilize the weight during growth in order to standardize the BMI at long term while ensuring growth and normal development. With the aim of reducing obesity and all its physical and psychosocial consequences, intervention with young and his family should first be the acquisition of healthy diet, regular practice of physical activities and the reduction of sedentary behaviour [13-15].
For many patients, it is preferable to start by reducing sedentary behaviours, and gradually add regular physical activity to promote fitness and well-being, without seeking only to burn calories. Reducing the bad eating and physical activity habits and work to improve the good ones is just as important. These approaches used alone or in combination will contribute to the acquisition of a healthy lifestyle and weight control in the short and long term. The success of the intervention, which is different for each individual, requires realistic and sustainable treatment strategies. It can be defined as a better quality of life, a greater self-esteem, a greater body image, higher energy levels and others [13-15]. For some patients, the prevention or slowing of weight gain may be the only realistic goal. On the other hand, the resumption of weight should not be regarded as a failure; it is rather a natural and expected consequence of a chronic health problem [13-15]. To ensure proper tracking, child and parents have interest to keep a copy of the objectives to be achieved. Finally, in the treatment of patient with T2DM, your chance of success will be considerably better if you could get a mutual agreement on a glucose target and on realistic weight loss goals.
Assist
It is important to assist patients and parents in their goals towards good management of T2DM and obesity of their child, because they have many obstacles to overcome, such as stress, lack of time, fear to be blamed, little or no access to recreational facilities and socio-economic factors [13-15]. To help them we need to transmit documentation adapted to the patient, to their interests and to the needs of the family. For example, you can provide them with a detailed diet model, respecting the dietary recommendations based on the energy needs of the child as well as an array of energy expenditure by physical activity based on the weight of the child, the intensity and the duration of physical activity such as the documents provided by the Canadian Society for Exercise Physiology (www.csep.ca).
Assist means also to help the child, and his entire family develop healthier habits, better ways of promoting the weight maintenance and glycemic control, such as daily weighing, glucose monitoring and the definition of realistic objectives, and to identify stimuli that may influence the success of interventions including people, situations, emotions that trigger eating unhealthy and those limiting physical activity in order to change them.
According to the American Association of Diabetes Educators (https://www.diabeteseducator.org/), there are many steps to educating those with T2DM. The main step is healthy eating as mentioned above, followed by being active. We should teach the patient and his family members different ways to incorporate exercise into their lifestyle such as going on family walk, taking more family outings that gets the family out of the home for hiking, walking, biking, swimming and others that are inexpensive alternatives to watching television. We should then teach the patient and his family about monitoring and taking medication. This would have to be under the supervision of the parents. Monitoring would include how to use a blood sugar glucose meter, knowing when to check the numbers and the meanings of these numbers, the target range, and how to record blood sugar levels. Monitoring should also include how to keep the patient’s food and physical activity journal especially at school. Any medications prescribed by the physician should also be monitored. I would stress the importance to patient and his parents how important it is to follow the regimen prescribed by the HCP. As discussed in the next section, to ensure that all the patient is adequately educate about all the aspects of his disease, you will arrange for him and his family to be seen by a T2DM specialized nurse.
The next step is problem solving which looks at situations in which the patient may struggle to stick to her new, healthy lifestyle. It consists of seven steps: 1) definition of the problem as accurately as possible; [2] clarification of the problem by putting it in its context (reformulate the problem can help in its clarification]; [3] collection of the greatest number of solutions [brainstorming]; [4] analysis of the impact of each proposed solution [weigh the pros and cons of each of the solutions]; [5] selecting the best solution or combination of solutions; [6] implementation of the selected solution [define the stages of implementation of this solution]; [7] evaluation of results and corrections of the different stages if necessary [13-15].
By using this approach to each problem experienced by the patient and his family, they become, with a little practice, more autonomous in the management of the problems affecting their T2DM and weight management goals [13-15]. Meetings with the child and his parents, it is also very important to find, or at least provide appropriate suggestions to address personal and family barriers as well as to assist in the development of the skills of family organization promoting the achievement of adequate behaviours. Finally, it is important to plan for the unexpected and relapses by providing information and resources [13-15]. If necessary, a psychology or psychiatry consultation may be necessary to help the patient to understand and solve psychological problems such as esteem self-esteem, body image, acceptance or depression problems related to the condition of the child [13-15]. All of these steps would not be included in one session as that is a lot to take in but handouts are available on the American Association of Diabetes Educators website. Also, I would talk with the patient and his family about enrolling in a diabetes camp in order to meet other kids in similar situations and hopefully help ease the adjustments into this new lifestyle, while finding support [13-15].
Education around lifestyle modification involves not just the child but also his/her family. The whole family may need education to understand the principles of treatment for T2DM and the critical importance of lifestyle changes if chronic complications are to be prevented. The whole family should be encouraged to change their diet consistent with healthy eating recommendations, including individualized counselling for weight reduction, reduced total and saturated fat intake and increased fibre intake [13-15]. The key areas that have been found important in children include elimination of sugar-containing soft drinks and juice; taking meals on schedule and in one place, with no other activity at the same time such as watching TV, and ideally as a family group; portion control by reducing portion sizes; and limiting high fat, high calorie density food in the home [13-15].
Exercise management means developing individual exercise programs that are enjoyable, affordable for the family, and participated in by at least one other family member. Families should be encouraged to develop an achievable daily exercise program, including reducing sedentary time. Opportunities may include using stairs instead of elevators; walking part of the way to school; using an exercise machine at home, or exercise DVDs; and walking with a family member after school [13-15].
Assist aims to intervene on the perception of children and parents, and to raise awareness of the predisposition to gain weight, the severity of the problem of weight and the benefits of treatment on health and quality of life [13-15]. Assist aims to discuss about self-esteem, body image and to learn how to decode the emotions that often push the kids to eat well beyond their needs. Assist is intended to help in the development of communication with the patient and in the development of parental skills as well as to increase knowledge about the regulation of body weight and in the control of glucose levels. Assist aims to understand what a healthy diet is and why it is necessary to increase physical activity and reduce sedentary behaviors to help in the management of T2DM. Assist aims to provide information on the ways to modify certain negative behaviours related to a mismanagement of body weight and glucose control. An essential aspect of the assist part of the “6As” model of counselling is to teach the patient and his parent how to establish appropriate goals in terms of weight loss, nutrition and physical activity and glucose targets [13- 15]. Finally, the following simple slogan to promote a healthy lifestyle among youth and their families: “5, 3, 2, 1, 0”, designating five [5] portions and more of fruits and vegetables per day, three [3] structured meals per day [including breakfast], two [2] hours or less of television or video games per day an [1] hour or more of moderate to vigorous physical activity daily and no [10] sugary drink is certainly a good toll to assist T2DM pediatric patients achieving their treatment goals.
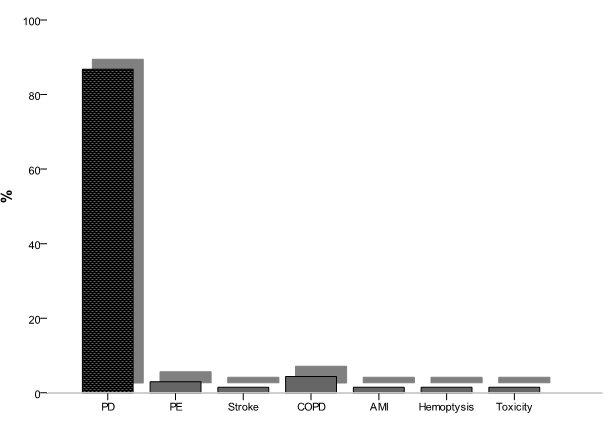
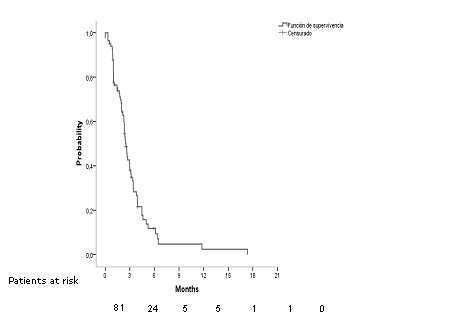
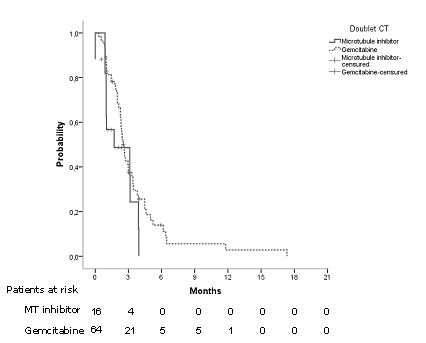
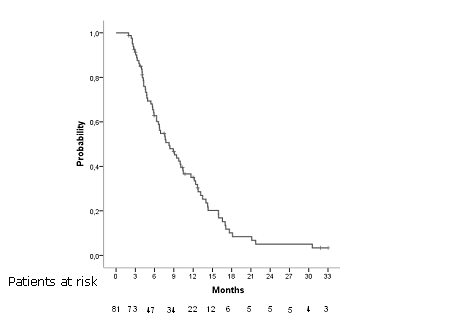
Arrange
Scientific evidences support the need for a multidisciplinary approach in the management of T2DM pediatric patients [13- 15]. They also confirm that a follow up with the family doctor, pediatrician and other HPC is essential to the acquisition and maintenance of glucose targets, weight loss goals and healthy habits [13-15]. This follow-up must be arranged with the patients and their parents, especially when other HPC or specialists are concerned, because in some cases, this can lead to unexpected financial expenses [13-15]. This monitoring can cause different emotions, especially when a medical specialist should be consulted. Given the chronic nature of T2DM, a long-term monitoring is essential, the “arrange” component of the “6As” model of counselling becomes an integral part in the monitoring of T2DM in pediatric patients. The frequency of meetings may vary depending on the patient and the treatment objectives [case by case] and must also be decided and discussed with parents, because this follow-up requires certain family organizations. The success of the treatment is directly related to the frequency of contacts, while the lack of follow-up or interventions can certainly lead to failure [13-15]. Arrange for follow-up may take the form of a brief phone call, from an email or a texto, or the use of a web applications or a return to the clinic, as needed.
Monitoring is essential to quickly identify any problematic situation, social or psychological aspects related to wrong eating or physical activity habits, which have repercussions and are responsible for failures to treatment. At meetings, it is important to discuss with the parents and the patient of what worked or not, to encourage behaviour which have been successful, in order to find solutions to improve or correct behaviour with little success and, above all, to support the family in its efforts, while establishing a good relationship with them to promote their returns if and keep them motivated and to provide additional support as needed [13-15].
The patient and his family should be educated on self-monitoring of blood glucose (SMBG). It is important to teach both the patient and his parents because the child are often too young and may need assistance until they get used to the system. SMBG is recommended with individuals with T2DM because it has been found to be very effective in controlling blood glucose levels. And for that you will need to arrange that the patient and his family be seen by a specialized diabetes nurse. Once T2DM is diagnosed, the patient will need to be followed regularly for early detection of complications. Once glycaemic goals have been met, the frequency of monitoring may be reduced to 2 to 3 fasting and 2 to 3 postprandial capillary checks per week. Clearly, children on insulin therapy should be undertaking more frequent testing. HbA1c should be assessed every 3 months if on insulin treatment.
Conclusion
HCP are in a very good position for supporting appropriate efficient intervention for the treatment of T2DM in pediatric patients. The reasons why the approach discussed above has more chance of success is that the work of behavior change is not taken on solely by the HCP. The main role for the HCP is to start sensitive conversations, achieve agreement on following through with effective treatment strategies, and support the patient in the initiatives that he or she undertakes. The 6As model of counselling comprises a manageable evidence-based behavioral intervention strategy that has the potential to improve the success of T2DM management with T2DM pediatric patients within primary care as this approach as already being successful for other metabolic disorders including pediatric obesity [13-15].
References
- Onge ES, Miller SA, Motycka C, DeBerry A (2015) A review of the treatment of type 2 diabetes in children. J Pediatr Pharmacol Ther 20: 4-16. [crossref]
- Jordan DN and Jordan JL (2012) Pediatric type 2 diabetes mellitus complications: a systematic review of the literature. J of Diab Res & Clin Met: 1-6.
- Reinehr T (2013) Type 2 diabetes mellitus in children and adolescents. World J Diabetes 4: 270-281. [crossref]
- Samaan MC (2013) Management of Pediatric and Adolescent Type 2 Diabetes. Int J of Pediatrics 1-9.
- Barnett T (2013) Type 2 diabetes mellitus: incidence, management and prognosis. Pediatrics and Child Health 23: 163-167
- Panagiotopoulos C, Riddell MC and Sellers AC (2013) Canadian Diabetes Association Clinical Practice Guidelines Expert Committee. Type 2 Diabetes in Children and Adolescents.
- Public Health Agency of Canada (2011) Chapter 5 – Diabetes in children and youth Diabetes in Canada: Facts and figures from a public health perspective.
- Amiel SA, Sherwin RS, Simonson DC, Lauritano AA, Tamborlane WV (1986) Impaired insulin action in puberty. A contributing factor to poor glycemic control in adolescents with diabetes. N Engl J Med 315: 215-219. [crossref]
- Kelsey MM, Zeitler PS2 (2016) Insulin Resistance of Puberty. Curr Diab Rep 16: 64. [crossref]
- Bailey CJ, Aschner P, Del Prato S, LaSalle J, Ji L, et al. (2013) on behalf of the Global Partnership for Effective Diabetes Management. Individualized glycaemic targets and pharmacotherapy in type 2 diabetes. Diab & Vasc Dis Res 10: 397–40.
- Hamman RF, Bell RA, Dabelea D, D’Agostino RB Jr, Dolan L, et al. (2014) The SEARCH for Diabetes in Youth study: rationale, findings, and future directions. Diabetes Care 37: 3336-3344. [crossref]
- Flint A and Arslanian S (2011) Treatment of Type 2 Diabetes in youth. Diabetes Care 34: S177-S183
- Plourde G (2014) Paediatric obesity: A guide on diagnosis, prevention and management.
- Plourde G (2014) Les Jeunes et l’Obésité: Diagnostics et Interventions. Les Presses de l’Université Laval.
- Plourde G (2013) Six As model of counseling in obesity. Can Fam Physician 59: 353. [crossref]
- Plourde G, and Marineau JM (2011) Reducing sedentary behaviours in obese patients: If they won’t exercise more – try to make them sit less. Parkhurst Exchange 63-71.
- American Diabetes Association (2011) Executive summary: standards of medical care in diabetes–2011. Diabetes Care 34 Suppl 1: S4-10. [crossref]
- Zeitler P, Fu J, Tandon N, Nadeau K, Urakami T, et al. (2014) ISPAD Clinical Practice Consensus Guidelines 2014. Type 2 diabetes in the child and adolescent. Pediatr Diabetes 15 Suppl 20: 26-46. [crossref]
- Plourde G (2011) Oral Diabetes medications: How to Personalize Therapy. Parkhurst Exchange. November/December 2011:50-53.
- Praveen PA, Kumar SR, Tandon N (2015) Type 2 diabetes in youth in South Asia. Curr Diab Rep 15: 571. [crossref]
- Van Buren DJ, Tibbs TL (2014) Lifestyle interventions to reduce diabetes and cardiovascular disease risk among children. Curr Diab Rep 14: 557. [crossref]
- McGavock J, Dart A, Wicklow B (2015) Lifestyle therapy for the treatment of youth with type 2 diabetes. Curr Diab Rep 15: 568. [crossref]
- TODAY Study Group, Zeitler P, Hirst K, Pyle L, Linder B, et al. (2012) A clinical trial to maintain glycemic control in youth with type 2 diabetes. N Engl J Med 366: 2247-2256. [crossref]
- Narasimhan S, Weinstock RS2 (2014) Youth-onset type 2 diabetes mellitus: lessons learned from the TODAY study. Mayo Clin Proc 89: 806-816. [crossref]
- Hoelscher DM, Kirk S, Ritchie L, Cunningham-Sabo L (2013) Academy Positions Committee. Position of the Academy of Nutrition and Dietetics: interventions for the prevention and treatment of pediatric overweight and obesity. J Acad Nutr Diet 113:1375-1394.
- Active Healthy Kids Canada (2012) Is Active Play Extinct? The Active Healthy Kids Canada 2012. Report Card on Physical Activity for Children and Youth, Toronto, Active Healthy Kids Canada.
- Pivovarov JA, Taplin CE, Riddell MC (2015) Current perspectives on physical activity and exercise for youth with diabetes. Pediatr Diabetes 16: 242-255. [crossref]
- Plourde G, Prud’homme D (2012) Managing obesity in adults in primary care. CMAJ 184: 1039-1044. [crossref]
- Fedewa MV, Gist NH, Evans EM, Dishman RK (2014) Exercise and insulin resistance in youth: a meta-analysis. Pediatrics 133: e163-174. [crossref]
- Nadeau KJ, Zeitler PS, Bauer TA, Brown MS, Dorosz JL, et al. (2009) Insulin resistance in adolescents with type 2 diabetes is associated with impaired exercise capacity. J Clin Endocrinol Metab 94: 3687-3695. [crossref]
- Rothman RL, Mulvaney S, Elasy TA, VanderWoude A, Gebretsadik T, Shintani A, et al. (2008) Self-management behaviors, racial disparities, and glycemic control among adolescents with type 2 diabetes. Pediatrics 121:e912e9.
- Wood JR, Silverstein J (2013) Incretins and amylin in pediatric diabetes: new tools for management of diabetes in youth. Curr Opin Pediatr 25: 502-508. [crossref]
- Jones KL, Arslanian S, Peterokova VA, Park JS, Tomlinson MJ (2002) Effect of metformin in pediatric patients with type 2 diabetes: a randomized controlled trial. Diabetes Care 25: 89-94. [crossref]
- Copeland KC, Silverstein J, Moore KR, Prazar GE, Raymer T, et al. (2013) Management of newly diagnosed type 2 Diabetes Mellitus (T2DM) in children and adolescents. Pediatrics 131: 364-382. [crossref]
- Inzucchi SE, Bergenstal RM, Buse JB, et al. (2012) Management of Hyperglycemia in Type 2 Diabetes: A Patient-Centered Approach Position Statement of the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care 35: 1364 -1379
- Rosenbloom AL, Silverstein JH, Amemiya S, Zeitler P, Klingensmith (2009) ISPAD Clinical Practice Consensus Guidelines 2009 Compendium. Type 2 diabetes in children and adolescents. Pediatric Diabetes 10: S17–32.
- Scheen AJ (2016) Precision medicine: The future in diabetes care? Diabetes Res Clin Pract 117: 12-21. [crossref]
- Dujic T, Zhou K, Donnelly LA, Tavendale R, Palmer CN, et al. (2015) Association of Organic Cation Transporter 1 With Intolerance to Metformin in Type 2 Diabetes: A GoDARTS Study. Diabetes 64: 1786-1793. [crossref]
- Dujic T, Causevic A, Bego T, Malenica M, Velija-Asimi Z2, et al. (2016) Organic cation transporter 1 variants and gastrointestinal side effects of metformin in patients with Type 2 diabetes. Diabet Med 33: 511-514. [crossref]
- GoDarts, Group UDPS, Wellcome Trust Case Control C, Zhou K, Bellenguez C, Spencer CC, et al. (2011) Common variants near ATM are associated with glycemic response to metformin in type 2 diabetes. Nat Genet 43:117–20.
- Reitman ML, Schadt EE (2007) Pharmacogenetics of metformin response: a step in the path toward personalized medicine. J Clin Invest 117: 1226-1229. [crossref]
- Scheen AJ (2014) Personalising metformin therapy: a clinician’s perspective. Lancet Diabetes Endocrinol 2: 442-444. [crossref]
- Shepherd M, Shields B, Ellard S, Rubio-Cabezas O, Hattersley AT (2009) A genetic diagnosis of HNF1A diabetes alters treatment and improves glycaemic control in the majority of insulintreated patients. Diabet Med 26:437–41.
- Pearson ER, Donnelly LA, Kimber C, Whitley A, Doney AS, et al. (2007) Variation in TCF7L2 influences therapeutic response to sulfonylureas: a GoDARTs study. Diabetes 56: 2178-2182. [crossref]
- Holstein A, Hahn M, Körner A, Stumvoll M, Kovacs P (2011) TCF7L2 and therapeutic response to sulfonylureas in patients with type 2 diabetes. BMC Med Genet 12: 30. [crossref]
- Sesti G, Marini MA, Cardellini M, Sciacqua A, Frontoni S, Andreozzi F, et al. (2004) The Arg972 variant in insulin receptor substrate-1 is associated with an increased risk of secondary failure to sulfonylurea in patients with type 2 diabetes. Diabetes Care 27:1394–8.
- Pearson ER (2016) Personalized medicine in diabetes: the role of ‘omics’ and biomarkers. Diabet Med 33: 712-717. [crossref]
- Kirchheiner J, Roots I, Goldammer M, Rosenkranz B, Brockmoller J (2005) Effect of genetic polymorphisms in cytochrome p450 (CYP) 2C9 and CYP2C8 on the pharmacokinetics of oral antidiabetic drugs: clinical relevance. Clin Pharmacokinet 44:1209–25
- Loganadan NK, Huri HZ, Vethakkan SR, Hussein Z (2016) Genetic markers predicting sulphonylurea treatment outcomes in type 2 diabetes patients: current evidence and challenges for clinical implementation. Pharmacogenomics J 16:209-219.
- Scheen AJ (2007) Pharmacokinetic interactions with thiazolidinediones. Clin Pharmacokinet 46: 1-12. [crossref]
- Kawaguchi-Suzuki M, Frye RF (2013) Current clinical evidence on pioglitazone pharmacogenomics. Front Pharmacol 4:147.
- Stage TB, Christensen MM, Feddersen S, Beck-Nielsen H, Brosen K (2013) The role of genetic variants in CYP2C8, LPIN, PPARGC1A and PPARgamma on the trough steady-state plasma concentrations of rosiglitazone and on glycosylated haemoglobin A1c in type 2 diabetes. Pharmacogenet Genomics 23:219–27.
- Avery P, Mousa SS, Mousa SA (2009) Pharmacogenomics in type II diabetes mellitus management: steps toward personalized medicine. Pharmgenomics Pers Med 2:79–91.
- Della-Morte D, Palmirotta R, Rehni AK, Pastore D, Capuani B, et al. (2014) Pharmacogenomics and pharmacogenetics of thiazolidinediones: role in diabetes and cardiovascular risk factors. Pharmacogenomics 15: 2063-2082. [crossref]
- Blüher M, Lübben G, Paschke R (2003) Analysis of the relationship between the Pro12Ala variant in the PPAR-gamma2 gene and the response rate to therapy with pioglitazone in patients with type 2 diabetes. Diabetes Care 26: 825-831. [crossref]
- Wang G, Wang X, Zhang Q, Ma Z (2007) Response to pioglitazone treatment is associated with the lipoprotein lipase S447X variant in subjects with type 2 diabetes mellitus. Int J Clin Pract 61:552–7.
- Yang H, Ye E, Si G2, Chen L, Cai L, et al. (2014) Adiponectin gene polymorphism rs2241766 T/G is associated with response to pioglitazone treatment in type 2 diabetic patients from southern China. PLoS One 9: e112480. [crossref]
- Bernington P, Chen J, Tibaldi F et al. (2011) LY2189265, a lobg-acting glucagon-like peptide-1 analogue showed a dose-dependent effect in insulin secretion in healthy subjects. Diabetes Obes Metab 13:434-438.
- Inge TH, Miyano G, Bean J, Helmrath M, Courcoulas A, et al. (2009) Reversal of type 2 diabetes mellitus and improvements in cardiovascular risk factors after surgical weight loss in adolescents. Pediatrics 123: 214-222. [crossref]
- Nadler EP, Reddy S, Isenalumhe A, et al. (2009) Laparoscopic adjustable gastric banding for morbidly obese adolescence affects android fat loss, resolution comorbidities, and improved metabolic status. J Am Coll Surg 209:638–644.
- O’Brien PE, Sawyer SM, Laurie C, Brown WA, Skinner S, et al. (2010) Laparoscopic adjustable gastric banding in severely obese adolescents: a randomized trial. JAMA 303: 519-526. [crossref]